Our approach
We value building strong and authentic relationships with our clients to deliver personalized and effective solutions.
At Promeditec, we collaborate with our clients to drive innovation in product development, production processes, and service delivery. Innovation and new technologies are at the core of our service philosophy and software projects.
We believe that innovation is essential for creating efficiency and effectively managing complex projects.
We believe that innovation is essential for creating efficiency and effectively managing complex projects.
INNOVATION
All Promeditec solutions and services are tailored to meet the specific needs of our clients.
With a high degree of customization, we ensure the most suitable and efficient solution for each individual project, supporting and working alongside our clients throughout the development and implementation phases.
With a high degree of customization, we ensure the most suitable and efficient solution for each individual project, supporting and working alongside our clients throughout the development and implementation phases.
TAILORED TO YOUR NEEDS
At Promeditec, our team of experts possess the knowledge and skills to analyze, design, and propose the most suitable solutions for any needs. By combining innovation, security, simplicity, and customization with experience and practicality, our team is able to deliver solutions with long-lasting added value.
EXPERIENCE
Promeditec solutions are designed to be simple, intuitive, and user-friendly. Our goal is to simplify the use of software for our clients, making the launch of even the most complex projects more manageable and successful. Promeditec’s innovative products place technology at the service of the customer, with the goal of eliminating the unnecessary and focusing on what is essential.
SIMPLICITY

Our services
Discover the range of services to meet your needs and empower your business.
CONSULTING
Guidance on innovative and efficient digital solutions to improve the efficiency and accuracy of clinical research.
E-LEARNING
Researching for the most suitable technology solutions, managing technical support activities, and organizing training activities for an effective e-learning experience: Masterclasses, Courses, Webinars, Live streaming, and Podcasts.
EVENTS
Organization and management of virtual, hybrid and on-site events supporting the client at all stages: from the consultancy to the live stage.
VIDEO PRODUCTION & POST-PRODUCTION
We work on the content to always deliver excellent audio/video quality, creating educational and creative video materials.
Your research,
our technologies
our technologies
Simplifying your clinical research with smart solutions.
Web platform to manage medical device approval documents and processes.
Electronic system to manage, store and track trial-related documents.
Implementation of clinical Databases and electronic-format Case Report Forms.
Web-based database for group clinical trials administrative management.

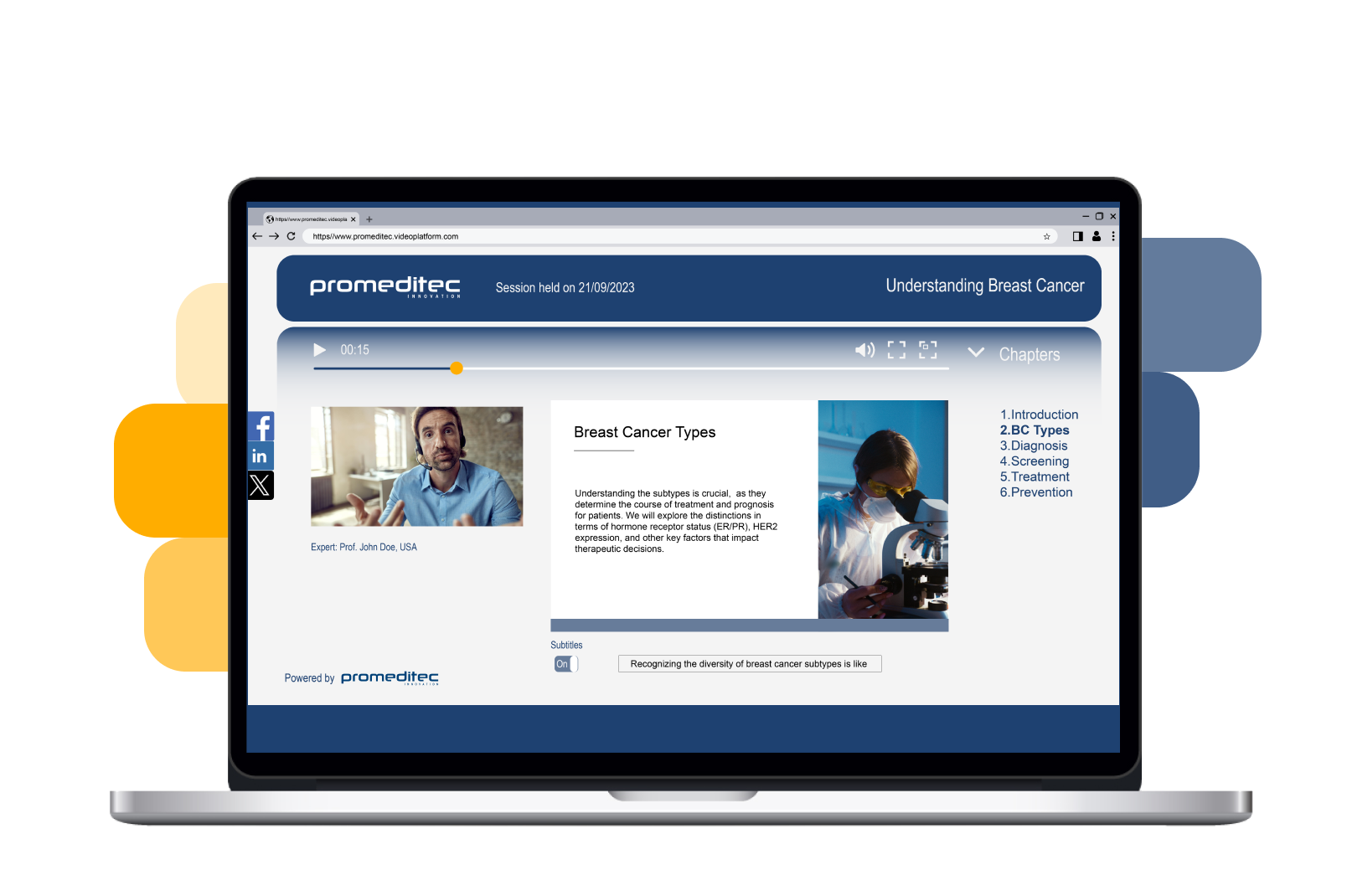
E-Learning Video Platform
Unlock the potential of online education with our customizable platform.
It provides a comprehensive solution for delivering and managing online learning materials, designed to offer engaging and impactful experiences.
It provides a comprehensive solution for delivering and managing online learning materials, designed to offer engaging and impactful experiences.